Water, the liquid commonly used for cleaning, has a property called surface tension. In the body of the water, each molecule is surrounded and attracted by other water molecules. However, at the surface, those molecules are surrounded by other water molecules only on the water side. A tension is created as the water molecules at the surface are pulled into the body of the water. This tension causes water to bead up on surfaces (glass, fabric), which slows wetting of the surface and inhibits the cleaning process. You can see surface tension at work by placing a drop of water onto a counter top. The drop will hold its shape and will not spread.In the cleaning process, surface tension must be reduced so water can spread and wet surfaces. Chemicals that are able to do this effectively are called surface active agents, or surfactants. They are said to make water “wetter.”
However, at the surface, those molecules are surrounded by other water molecules only on the water side. A tension is created as the water molecules at the surface are pulled into the body of the water. This tension causes water to bead up on surfaces (glass, fabric), which slows wetting of the surface and inhibits the cleaning process. You can see surface tension at work by placing a drop of water onto a counter top. The drop will hold its shape and will not spread.In the cleaning process, surface tension must be reduced so water can spread and wet surfaces. Chemicals that are able to do this effectively are called surface active agents, or surfactants. They are said to make water “wetter.”
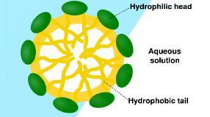 Surfactant molecules have the tendency to create micelles – spherical structures that can completely encapsulate a microscopic oil droplet so that it will be readily emulsified in an aqueous solution. Surfactants perform other important functions in cleaning, such as loosening, emulsifying sinkdishes(dispersing in water) and holding soil in suspension until it can be rinsed away. Surfactants can also provide alkalinity, which is useful in removing acidic soils.
Surfactant molecules have the tendency to create micelles – spherical structures that can completely encapsulate a microscopic oil droplet so that it will be readily emulsified in an aqueous solution. Surfactants perform other important functions in cleaning, such as loosening, emulsifying sinkdishes(dispersing in water) and holding soil in suspension until it can be rinsed away. Surfactants can also provide alkalinity, which is useful in removing acidic soils.
Surfactants are classified by their ionic (electrical charge) properties in water: anionic (negative charge), nonionic (no charge), cationic (positive charge) and amphoteric (either positive or negative charge).
Modern detergents contain more than surfactants. Cleaning products may also contain enzymes to degrade protein-based stains, bleaches to de-color stains and add power to cleaning agents, and blue dyes to counter yellowing. Like soaps, detergents have hydrophobic or water-hating molecular chains and hydrophilic or water-loving components. The hydrophobic hydrocarbons are repelled by water, but are attracted to oil and grease. The hydrophilic end of the same molecule means that one end of the molecule will be attracted to water, while the other side is binding to oil. Neither detergents nor soap accomplish anything except binding to the soil until some mechanical energy or agitation is added into the equation.
 Neither detergents nor soap accomplish anything except binding to the soil until some mechanical energy or agitation is added into the equation. Swishing the soapy water around allows the soap or detergent to pull the grime away from clothes or dishes and into the larger pool of rinse water. Rinsing washes the detergent and soil away. Warm or hot water melts fats and oils so that it is easier for the soap or detergent to dissolve the soil and pull it away into the rinse water. Detergents are similar to soap, but they are less likely to form films (soap scum) and are not as affected by the presence of minerals in water (hard water).
Neither detergents nor soap accomplish anything except binding to the soil until some mechanical energy or agitation is added into the equation. Swishing the soapy water around allows the soap or detergent to pull the grime away from clothes or dishes and into the larger pool of rinse water. Rinsing washes the detergent and soil away. Warm or hot water melts fats and oils so that it is easier for the soap or detergent to dissolve the soil and pull it away into the rinse water. Detergents are similar to soap, but they are less likely to form films (soap scum) and are not as affected by the presence of minerals in water (hard water).
Detergents may be made from petrochemicals or from oleochemicals derived from plants and animals. Alkalis and oxidizing agents are also chemicals found in detergents. Here’s a look at the functions these molecules serve:
Petrochemicals/Oleochemicals
These fats and oils are hydrocarbon chains which are attracted to the oily and greasy grime.
Oxidizers
Sulfur trioxide, ethylene oxide, and sulfuric acid are among the molecules used to produce the hydrophilic component of surfactants. Oxidizers provide an energy source for chemical reactions. These highly reactive compounds also act as bleaches.
Alkalis
Sodium and potassium hydroxide are used in detergents even as they are used in soapmaking. They provide positively charged ions to promote chemical reactions.

Suspendisse pulvinar, eros quis tempor ultricies, est augue gravida erat, at iaculis mi nibh ac est. Nulla tristique nibh ut lorem mattis euismod. Praesent aliquam suscipit rhoncus. Fusce purus arcu, ullamcorper sit amet ullamcorper eu, congue non arcu. Morbi vehicula arcu at arcu pellentesque at sollicitudin dolor sodales.
Praesent id tortor in lectus fermentum tincidunt. Nunc elit lectus, suscipit sed rutrum nec, malesuada a magna. Donec consequat pellentesque sem ac vehicula. Cras congue elit sollicitudin purus bibendum semper. Donec pulvinar sapien eu nulla hendrerit pellentesque. Pellentesque massa lectus, placerat a.
Morbi nec massa lorem. Morbi eu tellus sit amet arcu condimentum auctor. Vivamus tristique consequat rhoncus. Cras erat mauris, placerat non venenatis eu, ullamcorper ultrices magna. Cum sociis natoque penatibus et magnis dis parturient montes, nascetur ridiculus mus. Ut a elit vitae orci lacinia cursus ac eleifend tortor nunc.